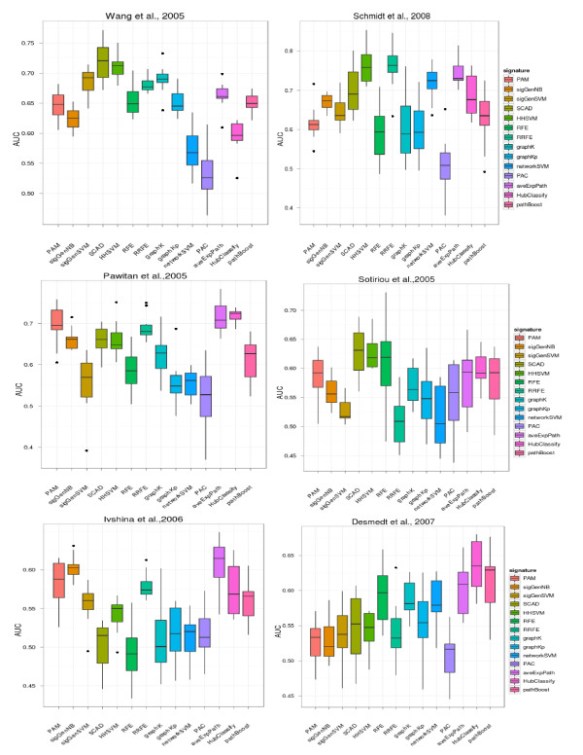
In our recent publication in BMC bioinformatics, we acompared a great deal of feature selection methods to finding prognostic biomakers in 6 breast cancer gene expresion data. No methods show significant performacne in prediction accuracy, feature selection stability and biogical interprety, which against previeous reseach results: current network-based appraoch did not show much benift in our analysis. Meanwhile, A group from NKI also show the simliar results in PloS One. The R codes for these algorithms in our paper is availiable as request.
Prognostic gene signatures for patient stratification in breast cancer – accuracy, stability and interpretability of gene selection approaches using prior knowledge on protein-protein interactions
Background
Stratification of patients according to their clinical prognosis is a desirable goal in cancer treatment in order to achieve a better personalized medicine. Reliable predictions on the basis of gene signatures could support medical doctors on selecting the right therapeutic strategy. However, during the last years the low reproducibility of many published gene signatures has been criticized. It has been suggested that incorporation of network or pathway information into prognostic biomarker discovery could improve prediction performance. In the meanwhile a large number of different approaches have been suggested for the same purpose.
Results
In this work we compared 14 published classification approaches (8 using network information) on six public breast cancer datasets with respect to prediction accuracy and gene selection stability. A gene set enrichment analysis for the predictive biomarker signatures by each of these methods was done to show the association with disease related genes, pathways and known drug targets. We found that on average incorporation of pathway information or protein interaction data did not significantly enhance prediction performance, but indeed greatly interpretability of gene signatures. Some methods (specifically network-based SVMs) could greatly enhance gene selection stability, but revealed only a comparably low prediction accuracy. Specifically network-based SVMs, performed well in terms of gene selection stability, whereas Reweighted Recursive Feature Elimination (RRFE) lead to very clearly interpretable signatures.
Conclusion
The results indicated that no single algorithm to perform best with respect to all three categories in our study. Incorporating network of prior knowledge into gene selection methods in general did not significantly improve classification accuracy, but greatly interpretability of gene signatures compared to classical algorithms.
A Critical Evaluation of Network and Pathway-Based Classifiers for Outcome Prediction in Breast Cancer
http://www.plosone.org/article/info%3Adoi%2F10.1371%2Fjournal.pone.0034796
Staiger C, Cadot S, Kooter R, Dittrich M, Müller T, et al. (2012) A Critical Evaluation of Network and Pathway-Based Classifiers for Outcome Prediction in Breast Cancer. PLoS ONE 7(4): e34796. doi:10.1371/journal.pone.0034796
Recently, several classifiers that combine primary tumor data, like gene expression data, and secondary data sources, such as protein-protein interaction networks, have been proposed for predicting outcome in breast cancer. In these approaches, new composite features are typically constructed by aggregating the expression levels of several genes. The secondary data sources are employed to guide this aggregation. Although many studies claim that these approaches improve classification performance over single genes classifiers, the gain in performance is difficult to assess. This stems mainly from the fact that different breast cancer data sets and validation procedures are employed to assess the performance. Here we address these issues by employing a large cohort of six breast cancer data sets as benchmark set and by performing an unbiased evaluation of the classification accuracies of the different approaches. Contrary to previous claims, we find that composite feature classifiers do not outperform simple single genes classifiers. We investigate the effect of (1) the number of selected features; (2) the specific gene set from which features are selected; (3) the size of the training set and (4) the heterogeneity of the data set on the performance of composite feature and single genes classifiers. Strikingly, we find that randomization of secondary data sources, which destroys all biological information in these sources, does not result in a deterioration in performance of composite feature classifiers. Finally, we show that when a proper correction for gene set size is performed, the stability of single genes sets is similar to the stability of composite feature sets. Based on these results there is currently no reason to prefer prognostic classifiers based on composite features over single genes classifiers for predicting outcome in breast cancer.